Imagine Re‑Running R&D—With AI at the Center
Imagine revisiting a pivotal drug development program from five years ago—this time with AI copilots embedded across research, trials, and compliance.
- How much faster could new compounds be identified?
- How many insights could be surfaced in real time from clinical data?
- How much sooner could life‑saving therapies reach patients?
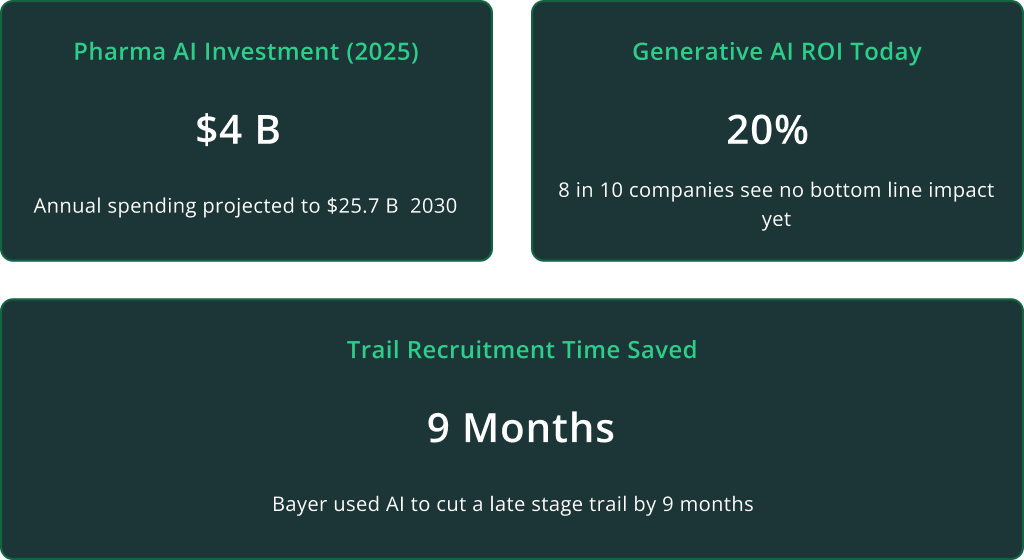
This isn’t a thought experiment anymore. Today’s AI capabilities are fundamentally reshaping how pharmaceutical R&D operates in compressing timelines, amplifying human expertise, and unlocking insights that were previously buried in data. Not surprising, global pharma AI investments soared to ~$4 billion in 2025 and are projected to reach $25.7 billion by 20301, reflecting huge ambitions. Yet many organizations have found that simply bolting AI onto existing processes yields little ROI. The winners will be those who reimagine legacy projects and workflows with an AI-first approach, capturing new value while their slower competitors fall behind.
The Past vs. Present: A Successful Transformation (Before AI)
Not long ago, digital transformation in pharma R&D meant modernizing legacy platforms, improving system performance, and eliminating manual bottlenecks. These efforts delivered meaningful gains—just without AI.
For example, a Fortune 500 pharmaceutical company partnered with Celestial Systems to modernize its electronic Trial Master File (eTMF) platform, the backbone for managing clinical trial documentation. The initiative introduced a modern, multi‑tenant cloud architecture, automated quality checks, and significantly improved system responsiveness, achieving dramatic gains in performance and usability while reducing operational friction.
The result was a faster, more reliable system that supported clinical teams at scale. Yet, by today’s standards, this transformation stopped just short of what is now possible.
Which raises a powerful question: What if AI had been part of that journey from day one?
Reimagining a Legacy R&D Platform—With AI in the Loop
Now fast-forward to today.
Reimagine that same eTMF platform augmented with AI copilots and intelligent automation. Instead of manually searching through thousands of trial documents, researchers and clinicians could ask natural-language questions and receive instant, context-aware answers from an AI trained on trial data. What once took hours or days could happen in seconds.
Machine learning models could proactively flag data quality issues, predict compliance risks before audits, and surface anomalies in real time. During development, AI coding assistants could accelerate platform enhancements and reduce time-to-implementation even further.
The original modernization delivered strong results. An AI-first approach would multiply its impact—not just by doing the same work faster, but by enabling entirely new ways of working.
This contrast—pre-AI success versus AI-powered reinvention—is playing out across the pharma R&D landscape today.
Where AI Creates the Greatest Impact Across Pharma R&D
Beyond a single platform or project, AI’s real value emerges when applied strategically across the R&D value chain. Several high-impact leverage points stand out:
1. Data-Driven Discovery & Preclinical Research
Early-stage R&D is inherently data-intensive. AI dramatically accelerates discovery by analyzing massive scientific, genomic, and chemical datasets in hours rather than months. Deep learning models can rapidly identify promising compounds, while generative AI can propose novel molecular structures that expand the innovation pipeline.
Strategic outcome: Faster time-to-discovery, broader pipelines, and improved odds of identifying breakthrough therapies ahead of competitors.
2. Smarter Clinical Trials & Development
Clinical trials are among the most expensive and time-consuming phases of R&D. AI enables smarter trial design, optimized patient recruitment, and advanced analytics that reduce delays and costs. Some organizations have already demonstrated that AI-driven recruitment optimization can shave months off late-stage trials.
AI-powered simulations and “digital twin” models further allow trials to be run more efficiently with fewer participants—without compromising outcomes.
Strategic outcome: Shorter trial timelines, reduced development costs, and faster time-to-market for new therapies.
3. AI-Enhanced Knowledge Management & Compliance
In pharma, documentation and compliance are as critical as the science itself. Generative AI copilots can read, summarize, and synthesize thousands of pages of research, clinical data, and regulatory literature in seconds—giving decision-makers with immediate clarity.
AI is also being used to accelerate pharmacovigilance, regulatory reviews, and clinical reporting, reducing manual effort while maintaining high accuracy.
Strategic outcome: Higher productivity, improved quality, faster regulatory submissions, and reduced compliance risk—freeing teams to focus on innovation rather than paperwork.
4. Post‑Market Safety & Pharmacovigilance
AI does not stop only at approval. Intelligent systems can continuously monitor real-world safety data, automate adverse event processing, and identify risk signals far faster than traditional approaches.
Strategic outcome: Improved patient safety, stronger regulatory trust, and lower long-term compliance costs.
What Executives Should Focus On: Outcomes, Not Algorithms
Across each of these areas, the value of AI is not about models or code—it’s about business impact:
- Faster insights and decision-making
- Reduced cycle times across R&D
- Lower costs and operational risk
- Greater innovation velocity
AI doesn’t just make existing processes faster—it enables new capabilities, such as real-time risk management and proactive decision support, that weren’t possible before.
Identifying Which R&D Processes Are Ripe for AI Reinvention
For leaders looking to move from experimentation to impact, a few guiding principles can help identify the right starting points:
- Target data-heavy workflows where teams are overwhelmed by documents, research, or trial data.
- Prioritize repetitive, manual processes that consume significant time and resources—AI excels at augmenting these.
- Focus where speed creates competitive advantage, especially steps that directly shorten R&D timelines.
- Assess data readiness and governance, ensuring AI augments human judgment responsibly in a regulated environment.
The goal is not unchecking automation, but thoughtful augmentation by pairing AI’s scale and speed with human expertise.
The Imperative for an AI‑First R&D Mindset
Pharma organizations that embrace an AI-first approach to reimagining R&D are positioning themselves for lasting competitive advantage. Faster discoveries. More efficient trials. Smarter decisions. And more time for scientists and clinicians to focus on what matters most, innovation and patient impact.
The full value of AI is unlocked not through isolated pilots, but through bold redesign of how work gets done. As industry observers have noted, organizations that truly redesign their operating models with AI will outpace those that merely tinker at the edges.
The future of Pharma R&D is already taking shape. The question is who will lead it?
If you’d like to explore these ideas further, join our monthly, Celestial Systems Open House – A chance to meet our team, see our latest solutions in action, and discuss how AI can be applied in your own organization.
We’d love for you to join the conversation.

